- Home
- Protein Products
- Biotinylated Protein Product List
- Biotinylated Proteins
Biotinylated Proteins
Carna' s biologically produced biotinylated proteins, exclusively developed and produced in-house, have a single biotin attached to the N-terminus (some to the C-terminus) and guaranteed purity >80%. These proteins are useful for the study of compound binding affinity and other protein-molecule interactions using Surface Plasmon Resonance (SPR), BioLayer Interferometry (BLI) or other assay formats that utilize biotin streptavidin interaction for detection. Its lower rate of interference with cellular proteins leads to the rapid acquisition of reliable data with high sensitivity and low background. Our Biotinylated Proteins are easily mobilized onto sensor surfaces without impairing their structure and activity.
Carna' s single-site, specifically biotinylated proteins are also useful for assay development using FRET, ELISA, AlphaScreen™, and HTRF® technologies.
Advantages of Carna's biologically biotinylated proteins
- Proteins are labeled with a single biotin at the N-terminus
- Easy-to-use ; no additional labeling required
- Native, catalytically active kinase domains are preserved for kinases
- High quality human proteins produced via Baculovirus expression system
- Stable activity determined post expression for kinases
- Select kinases available pre-activated (via ATP treatment) and non-activated (without ATP treatment)*
* products with (*) are available in both activated and non-activated forms as described
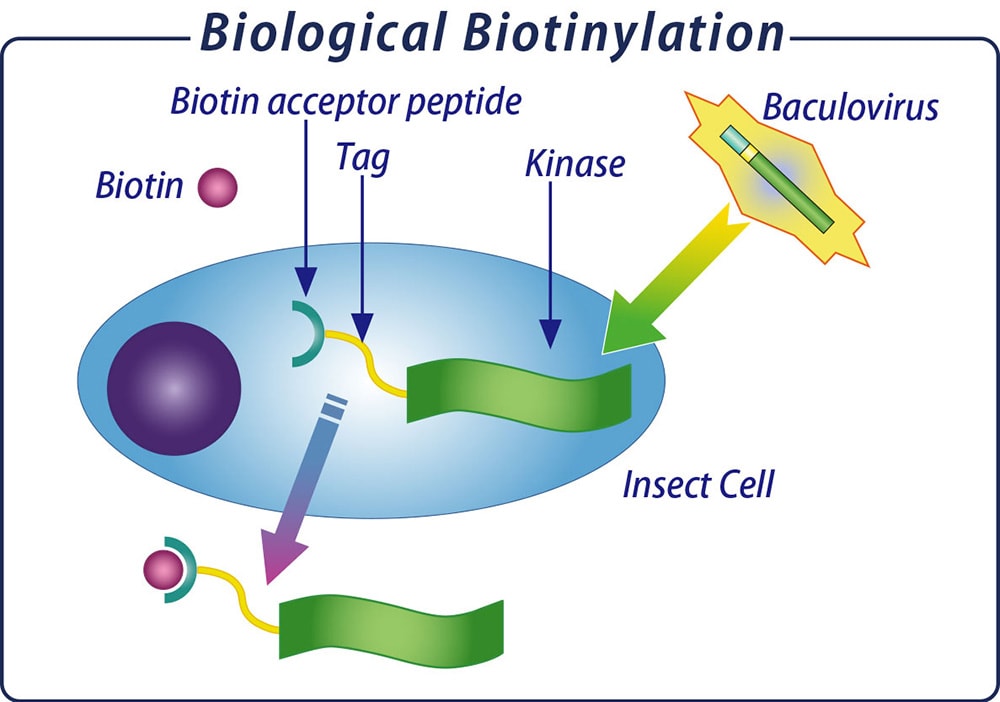
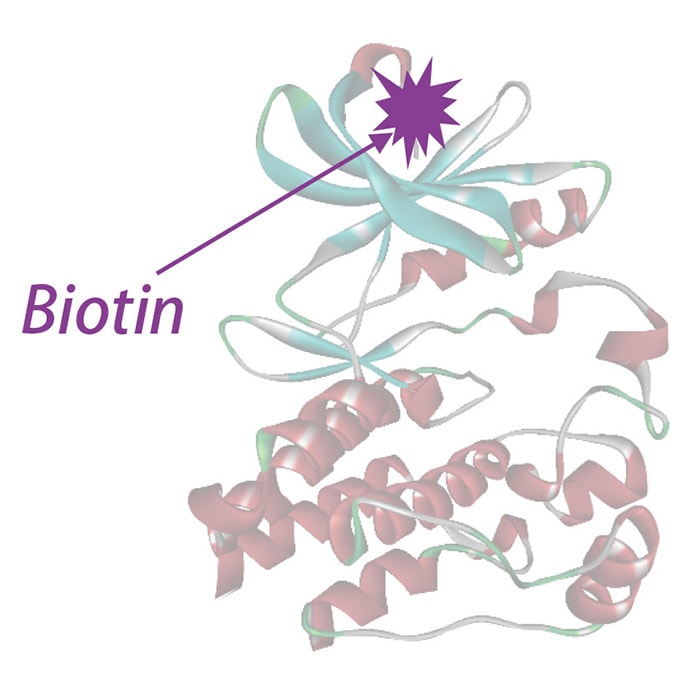
Carna' s single-site specifically biotinylated kinase
(Note)Chemical Biotinylation Disadvantages
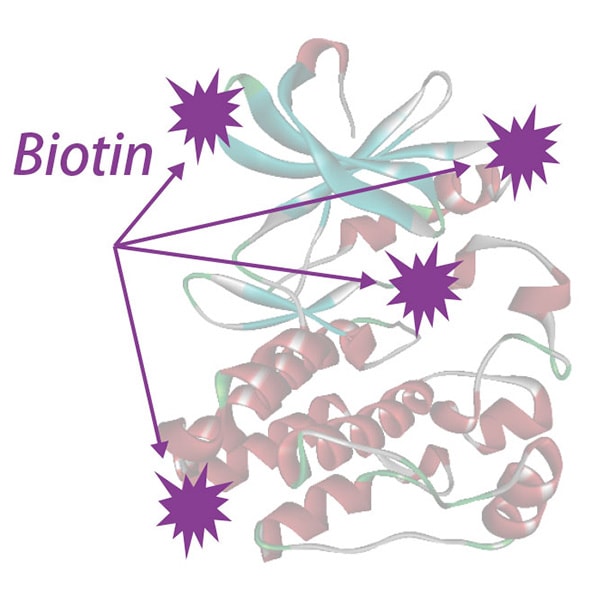
Chemically biotinylated kinase
- Multiple biotins may interfere with the ATP binding site following chemical biotinylation
- Kinase activity and structure can be altered after chemical modification
- Additional purification may be required (to eliminate non-reacted biotin)
High affinity between biotin and streptavidin
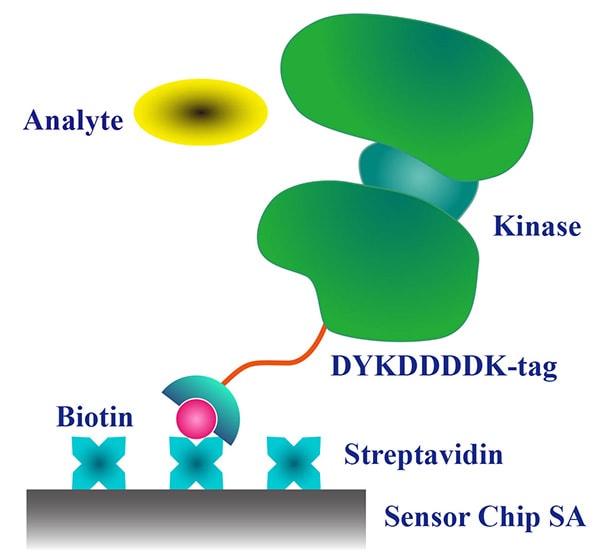
The strong binding between streptavidin and biotin is an invaluable asset that is widely utilized in biochemical experiments. Numerous applications including evaluation of intermolecular interactions based on the surface plasmon resonance (SPR) detection ( image on the right), determination of on-off rates and binding kinetics are made easier using Carna's biotinylated proteins.
SPR data examples* using Carna’s Biotinylated Proteins are shown on the list page.
*Measured using Biacore T200 in collaboration with Oncolines B.V.
